Using digestibility values in ration formulation
Joanne Siciliano-Jones, F.A.R.M.E. Institute Inc.
Most nutritionists depend on an energy value of some type to balance dairy rations. Unfortunately, predicted energy values for forages are frequently suspect. In two recent reviews, the problem of overestimation of energy values was discussed (Smith et al., 1996 and Weiss, 1996). Diets based on corn silage were identified as having the most severe overestimation of energy content. Thus we see discounting of forage energy values in the field. In extreme cases the NEl value of a ration is ignored entirely in favor of another indicator of energy content such as non-structural carbohydrate level. With experience, this "seat of the pants" approach can be made to work in many herds. Problems, in the form of poor cow performance and lost accounts, begin to surface with inexperienced personnel and in "abnormal" years or situations. Improving the accuracy of energy (NEl or ME) estimates for feed ingredients is critical to solve this problem.
Nutrient digestibility is one of the key components in determining energy available for milk production (NEl or ME). A quick review of the relationship between gross energy (GE) and net energy for lactation (NEl) reminds us that digestibility (fecal loss) is the first correction made to GE values, as they are discounted to reflect NEl. This presentation will focus on using digestibility values to more accurately predict the energy content of dairy rations.
At the present time, two methods are available to dairy nutritionists who would like to better account for digestibility differences between and within feedstuffs. These are the Ohio State Energy equation and the Cornell Net Carbohydrate and Protein System (CNCPS). The Ohio State Energy equation fragments feedstuffs into ideal digestion pools, based on chemical analysis. The energy content of the feed is then determined by applying standard digestibilities to these pools. This approach has resulted in the OSU energy equation being a better predictor of energy availability than the older ADF-based energy prediction equations. The CNCPS takes a similar approach, although it is executed in a more complex, dynamic system. For the CNCPS, digestibility is described both in terms of rate and extent of digestion. Furthermore, the CNCPS allows the user to adjust digestibility parameters if desired.
Determining feedstuff digestibility
The determination of feedstuff digestibility is not a simple task. Unlike many routine feedstuff analyses, there is no single, recommended procedure for digestibility analysis. Nutrient digestibility may be determined in vivo (in the animal), in situ (in place), or in vitro (in glass). In addition, digestibility of feeds can be described for either the total digestive tract or its various digestive compartments. To complicate matters further, digestibility determinations frequently differ depending upon which part of the digestive tract is to be simulated.
There should be no question that in vivo tests are the "gold standard" for digestibility testing. Because of the variation in digestibility between cows, in vivo tests require a substantial number of animals, and obviously a large amount of the feed to be tested. These requirements make in vivo testing impractical for application in balancing rations. In addition, testing digestibility in high producing cows and evaluating the effects of other dietary components is problematic due to the fact that only the feed to be tested can be fed during the digestibility test.
Since in situ and in vitro tests can be conducted with small quantities of a feedstuff, these tests may be used on a routine basis to provide digestibility values for ration balancing. These tests have lower inherent variability than in vivo tests and so require fewer replications to produce a reliable value. Both in vitro and in situ digestibility tests have been developed to mimic ruminal digestion. In vitro and in situ digestibility testing methods are subject to a number of factors which can affect both their accuracy and precision. Only factors which can impact the application of these methods in ration balancing will be discussed here. A detailed discussion of factors affecting accuracy and precision in digestibility testing can be found in a recent review by Weiss (1994).
In vitro tests consist of setting up an artificial digestion system. The most common system in the US is the Tilley and Terry in vitro system with simulates ruminal digestion. Several modifications and enhancements have been made to the original method but the basic procedure remains the same. The artificial digestion system consists of an incubation system such as a shaking water bath to maintain a constant temperature of 39 C, vessel(s) containing a buffer solution and rumen fluid, and the test feed. The system is usually supplemented with a nitrogen source such as ammonium bicarbonate and, in the original system, vitamins and minerals. The test feed is dried and finely ground through a 1 or 2 mm screen. The resulting system is a close approximation of the ruminal fermentation, although most labs are unable to maintain a protozoal population. Because the fermentation is fueled only by the feedstuff to be tested and some inorganic nutrients, using this type of system to evaluate digestibility of non-forage ingredients may not be appropriate.
The Tilley and Terry in vitro digestibility test has several advantages. Tests can be conducted with only a very small quantity (.5 grams) of material. Both rates and extents of digestion can be determined. The most striking advantage for the Tilley and Terry system is its degree of precision. Coefficients of variation in the range of one to two percent are readily attainable. Much of the precision in this system can be attributed to the use of finely ground test samples, which reduces sampling error. Because in vitro digestibility tests typically report extent of digestion, one of the best uses of this information is to predict TDN or NEl. A TDN value can be calculated directly from in vitro results. Alternatively, in vitro NDF digestibility (extent) can be entered into the OSU energy equation in place of the "average" value which is currently used. This should improve the accuracy of the equation for predicting energy content of forages, in particular.
For application in the ration balancing, Tilley and Terry in vitro digestibility tests can be faulted on three counts. First, the system is not designed to provide information about digestion of soluble components of feedstuffs. The second is that this is an artificial system which is highly buffered and not subject to the same pattern of pH fluctuations as is the rumen of a lactating cow. The test feed is also isolated from the effects of the high starch and/or fat levels found in the diet of the high producing cows whose digestive function is to be predicted. Accuracy of in vitro digestibility determinations is dependent upon the diet of the animal from which the ruminal fluid is obtained (Weiss, 1994). The final problem with this system relative to predicting digestion of forages in high producing cows is the use of finely ground samples. Finely ground feeds are known to be more digestible than coarsely ground or unground materials. For a heterogeneous feed like corn silage, this is especially problematic because the increase in digestibility with grinding is not uniform within a given corn silage and definitely not between corn silages. The possibility of inconsistent digestibility responses to grinding poses a serious problem for nutritionists attempting to incorporate digestibility data in their ration balancing system.
An automated in vitro system (AIV) has been developed by researchers at Cornell to determine rates of digestion with minimal labor requirement. This system determines nutrient disappearance by measuring gas production in a Tilley and Terry type ruminal in vitro system. Because gas evolution rather than disappearance of a residue are being measured, the AIV procedure also allows digestibility of soluble nutrients to be measured. The AIV system still suffers from the remaining two drawbacks associated with other Tilley and Terry type in vitro systems; test feeds are finely ground and fermentation occurs under highly controlled, artificial conditions. In addition, the AIV method has a very unique problem associated with the relationship between gas evolution and fermentation pattern. Gas is measured in this system according to volume without correction for the ratio between methane and carbon dioxide. Methane and carbon dioxide differ in molar volume. Further, the ratio between these two gases changes based on the fermentation substrate, particularly the forage:concentrate ratio. These facts raise the possibility that the AIV procedure may not be accurate enough to describe corn silage digestibility for ration balancing.
A quite different type of in vitro digestibility test is to use commercially produced enzymes or enzyme cocktails to predict digestibility of feedstuffs. This technique is more common in Europe than in the US. One advantage is consistency. These types of systems use purified enzymes and strictly defined test conditions, removing concerns about possible variation in the rumen inocula. This type of system may also be designed to simulate intestinal digestion. However, use of purified enzymes may also be a disadvantage, as the enzymes used to simulate ruminal digestion are generally derived from fungal organisms and may not accurately mimic the action of ruminal enzymes.
The in situ technique was developed in part to address the concerns about in vitro digestibility tests being conducted in a highly controlled, artificial environment. With the in situ technique, the test feed is weighed into a cloth (dacron) bag with a defined porosity calculated to minimize loss of feed dry matter while allowing adequate microbial entry to produce normal digestive activity. The dacron bag containing the test feed is sealed and then incubated in the rumen of a cannulated animal. Extent of digestion can be measured using a single incubation time which is equal to the expected ruminal retention time of the feedstuff; multiple incubation times can be used to develop rates of digestion. Test feeds can be evaluated in the as fed form, although some labs routinely grind test samples. The major advantage of in situ digestibility evaluations is the ability to more closely simulate the system (the reticulo-rumen) which is being described. In situ testing offers the opportunity to evaluate digestibility of a feedstuff in the form in which it is to be fed. Ellis et al. (1994) have reported that reducing forage particle size by grinding can reduce or remove digestion time delay, leading to their recommendation to estimate digestion rates of feed particles in a form similar to that entering the rumen. Furthermore, the feedstuff being tested is exposed to the normal diurnal fluctuations in ruminal pH and any potential associative effects resulting from normal protein and energy supplementation. The fact that in situ digestibility data incorporated into a kinetic model resulted in close approximations of in vivo digestion (Moore et al., 1989) suggests a distinct advantage in using in situ digestibility data for ration balancing.
In situ digestibility determinations have one major disadvantage relative to the in vitro tests. In situ forage digestibility determinations have a higher variability than do in vitro tests. Coefficients of variation for in situ forage digestibility evaluations will normally be in the range of 3-5%. Finely ground feedstuffs generally have coefficients of variation for in situ digestibility that are no higher than typical coefficients of variation in vitro. While grinding forage samples will increase the precision associated with in situ digestibility testing, accuracy of the digestibility prediction is likely to suffer. A caution about in situ digestibility testing relative to accuracy of the test is in order. The animal used in the in situ test must be similar both with regards to physiology and feeding program to the animal for which the ration is to be formulated. In situ digestibility results are sensitive to forage:grain ratios and stage of lactation of the animal.
In spite of these potential problems, with careful quality control it is possible to reduce variability in in situ digestibility testing to acceptable levels while maintaining the advantage of conducting a digestibility test under conditions similar to those encountered by the feedstuff when it is fed. It has been the experience of the authors that using in situ digestibility data in a dynamic model can help explain poor milk production in well fed, properly managed herds with no overt barriers to increased production. More importantly, in situ digestibility data can be used to formulate rations which correct poor production problems. The best use of in situ digestibility data is in a model which utilizes information on both the rate and extent of digestion.
The in situ model
Feeds entering the rumen can go through one of two possible routes: they can be (1) degraded or (2) passed to the lower tract. In some sense, the two processes compete against one another: an increase in passage results in a decrease in degradation. Since digestion of a nutrient is proportional to degradation rate and inversely proportional to passage rate. These relationships form the foundation of the equation used to predict nutrient digestibility:
Digestibility coefficient = kd/(kd + kp),
where kd is rate of digestion
and kp is rate of passage
Rate of digestion, kd, is determined by measuring nutrient disappearance in situ at a number of time points. The estimation of kp, the rate of passage, is done through an independent procedure (e.g., markers). Because of the cost and difficulty involved in directly determining passage rate, standard values are often used. One must realize that the rate of passage is as important to the calculation of the digestibility coefficient as is the rate of degradation, but that the in situ procedure does not yield any information on the rate of passage.
The above equation is applicable only to a feed made of one uniform component. Generally, this is not the case since feeds are made up of various fractions (e.g., protein, fat, various carbohydrates, etc.). Even within a nutrient category (e.g., protein), a feed will contain various sub-groups, called pools. Because of this, the model needs to be expanded to accommodate multiple, independent and uniform sub-groups. Typically, we would assign up to four pools, with the pools defined by degradation characteristics. Pool A is assumed to be instantly degradable, pools B1 and B2 are both potentially degradable but degrade at different rates and pool C is completely undegradable. With a multi-pool example, digestibility coefficients would be calculated for each pool independently, allowing for the use of differing rates of passage for each pool. Total digestibility of the nutrient within the feed would then be calculated by weighted average of each pool’s digestibility coefficient.
Variation among forages in in situ digestibility
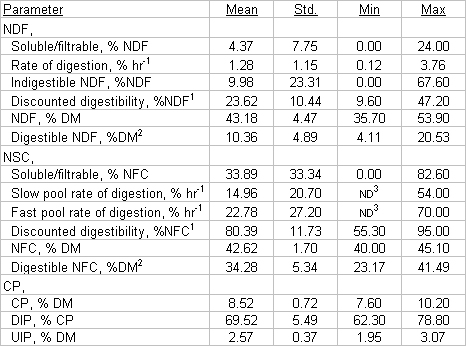
It is not surprising that different species of forages differ in rate and/or extent of digestibility. At F.A.R.M.E. Institute 1996 average in situ NDF digestibility (discounted for a 3%h-1 rate of passage) has ranged from 27.7% for corn silage to 37.3% for orchardgrass silage (J. Siciliano-Jones, unpublished data). Alfalfa silage NDF digestibility fell between corn silage and orchardgrass with an average value of 30.2%. The surprising finding has been the degree of variability for in situ digestibility within a forage type. Variation in digestibility observed in on farm samples is the result of the combined effects of forage genetics, environmental conditions, harvest timing, and harvest and ensiling management. To illustrate the variability encountered in the field, we present data collected by F.A.R.M.E. Institute using a single methodology for one crop, corn silage.
In evaluating the digestibility data presented here, it is important to note that most of the samples tested are from herds experiencing lower than normal milk production. In herds which are not experiencing problems in milk production, digestibility data is quite similar to the information found in the CNCPS.
Corn silage samples from the 1996 season had similar digestibility kinetics to those determined in 1995 samples. The one exception to this statement is NFC digestibility. More samples with hard, physiologically mature kernels were received in 1996, resulting in lower NFC digestibility. Another difference appeared to be related to kernel processing. Kernel processed samples generally have much higher proportions of NFC which end up in the soluble/filtrable pool. The NFC remaining in the potentially digestible pool has generally been less rapidly digested than was observed in 1995.
For most of the problem herds in the past 2 years, poor NDF digestibility has been a major contributor to the problem. Digestibility of NDF in Table 1 helps illustrate this issue. The average NDF digestibility was 23.6% of the NDF after correction for passage out of the rumen. With the observed values for chemical composition and NFC digestibility in these samples, it can be calculated that NDF digestibility must be approximately 35% of the NDF to result in a TDN value of 69% (converts to an NEl of .72 mcal/lb.). With the current emphasis on breeding corn hybrids for high digestibility, many producers are puzzled by these results. What causes these problems? Were hybrid genetics misrepresented? The answer is NO.
In fact, the digestibility values observed on a given farm are the net result of combined effects of management, environment, and hybrid genetics. Just as it is foolish to expect a well bred Holstein cow to produce up to her genetic potential for milk under poor management conditions, even the most digestible corn silage hybrid lose digestibility due to growing conditions or poor harvest and ensiling management. One of the most critical determinants of digestibility is maturity of the crop. Fortunately, this problem is manageable and well recognized among producers. Another digestibility determinant is growing environment, especially soil moisture. A negative relationship between in vitro digestibility and soil moisture has been shown in a two site, two year study in New York (Van Soest, 1996). Anecdotally, we have observed similar relationships with in situ testing of farm forage samples. The results shown in Table 2 were obtained from three farms in Eastern NY which differed in soil drainage; farm 1 had the best drained soil and farm 3 had the worst drainage. Both NFC and NDF digestibility were affected in these examples; however, it is more common to find poor NDF digestibility only when soil moisture is high.
From Tables 1 and 2, the high level of variability in digestibility parameters is evident. Since digestibility has an enormous impact on energy content of feedstuffs, it only makes sense to try to account for these differences in digestibility. Describing digestibility in terms of both rate and extent of digestion helps to more fully account for the impact of digestibility changes on the energy status of the animal. The importance of determining the rate of digestion lies in the fact that the rumen is not a closed system. Digesta particles flow constantly out of the rumen, whether digestion is complete or not. However, some researchers believe it is more appropriate to merely describe digestibility pools known to differ in rate of digestion and then assume a "standard" digestion rate for each pool. This may be a less expensive approach, but the important question is whether this approach will allow us to accurately estimate energy available to the animal.
What to measure: pools or rates?
The debate continues as to what is most important to measure: pools or rates. Obviously, efforts would be most effective if targeted to measuring that parameter which has the largest effect on the degradability estimate, that is, the parameter which has the largest marginal effect on degradability. This is best explored with calculus as in Siciliano-Jones and St. Pierre (1997), but will be covered in briefer fashion here. The importance of passage rate in determining the digestibility coefficient was discussed above. The relative importance of passage and digestibility in that equation change. When digestion rate is faster than passage rate, passage rate is the more important component. However, when digestion rate is slower than passage rate, digestion rate becomes the more critical component of the equation. For forage fiber digestion, digestion rate is generally slower than passage rate which makes rate of digestion the most critical measure.
For pool sizes, the relationships are not as straight-forward. This is because the pool sizes are not independent since their sum must be equal to 100. As a result, nothing can be said on the relative importance of determining pool sizes unless their variances and covariances, within each feed type are known. Therefore, the relative influence of pool size(s) versus degradation rate(s) is feed specific and no general conclusion can be drawn. For a given feed, the relative importance of each requires an estimate of the mean for each parameter and their possible range (variance).
To explore this area further, three forages were selected from the feed library of the CNCPS. The selected forages were; corn silage with 50% grain, a mixed alfalfa/grass silage with 15% CP and 52% NDF, and a grass silage with 20% CP and 48% NDF. Only fiber (NDF) digestibility was evaluated. Indigestible fiber as calculated from lignin content and rate of fiber digestion were the parameters selected for study. Both parameters were increased or decreased by 20% and the change in metabolizable energy content, as calculated by the model, was noted. The results are presented in Table 3.
This exercise suggests that changes in rate of NDF digestion have an effect on the energy available to the animal that is equal to or greater than the effect of changing indigestible fiber content. However, expressing variability of NDF digestion parameters in Table 1as the standard deviation divided by the mean leads to the conclusion that indigestible NDF may be more variable than rate of digestion. Therefore, a clear-cut answer to the question of which measure is more critical requires further study.
Taking a different approach to the question of whether pool size or rate is most critical in describing a feedstuff leads to the question of practicality and offsetting errors. In fitting a degradation curve to a limited series of in situ disappearance data, it is quite possible to underestimate (or overestimate) indigestible NDF. The larger the data series, the more accurate our estimates of digestion parameters. Unfortunately, cost also increases rapidly. As a result, we must search for a compromise between precision and cost. On a practical basis, it is important to remember that very high levels of precision may not be worth the added cost. The old saying about measuring with a micrometer and cutting with an ax comes to mind.
Because of offsetting errors, when indigestible NDF is underestimated (or overestimated) rate of digestion is overestimated (or underestimated) proportionately because the curve is being fit to describe a particular amount of material which is digested in a given period of time. Since digestion at any point in time is a product of rate and extent of digestion, any error in estimation of pool size (extent) must be offset by a change in the rate of digestion. The following example serves to illustrate this issue.
For a forage which has an NDF digestibility of 71.7% at 48 h, several combinations of rate and extent values are possible solutions. In scenario 1, we will assume a forage with digestibility characteristics similar to those found in the CNCPS for corn silage. In this example, indigestible fiber equals 25% of the NDF and rate of digestion for digestible fiber is 3.61% h-1. If we completely miss detecting the indigestible fiber fraction (indigestible fiber = 0) in scenario 2, we can estimate that the rate of digestion for the digestible fiber will be 2% h-1, using the relationship described in equation (4) above. The result is two very different descriptions of fiber digestion parameters. After adjusting for rate of passage, we can estimate just how much impact these different descriptions have on digestible NDF available to the animal. Calculating digestible NDF with an assumed rate of passage of 3% h-1 using equation (5) above, scenario 1 results in a digestible NDF value of 40% of the NDF while scenario 2 results in a value of 40.9%. This is a coefficient of variation of 2.2% which is well within the acceptable range for laboratory measures. A higher rate of passage would be expected to magnify the differences between scenarios 1 and 2. Assuming a rate of passage of 4% h-1 which would be quite reasonable for a forage fed to a high producing cow, scenario 1 gives a digestible NDF content of 33.3% while scenario 2 results in a digestible NDF of 35.6%. These values are not very different numerically. The difference in values results in a coefficient of variation of 6.7% which is slightly high for laboratory measures (5% is generally accepted), but probably acceptable for a biological measurement to be applied in a practical setting. As the field of digestibility testing to support ration balancing progresses, it is possible that compromises between precision and cost will no longer be required.
In situ digestibility data in ration balancing - a practical example
In essence, using in situ digestibility data to balance a ration is no different than using traditional forage tests. The goal is to describe the forage, since this appears to be the most variable component with regards to digestibility data, and adjust the other ration components to make up for any deficiencies in the forage. While this has not happened in the authors experience, it is also possible that higher than normal digestibility could result in the forage replacing higher priced ingredients. The difficulty we encounter is that digestibility is a dynamic measure which is influenced by rate of passage. As a result, we need to use a system such as the CNCPS which takes rate of passage and rate of digestion into account in calculating digestible nutrients available to the cow.
Over the past two years, in situ digestion parameters generated by F.A.R.M.E. Institute have been used in approximately 100 herds to adjust ration formulations. In almost all cases, milk production improved between 3 and 10 lbs. per head per day.
The following example illustrates the concept of using in situ digestibility data in ration balancing and the type of results which are possible. The herd described here was experiencing poorer than normal milk production. Much of the problem was occuring after peak production; cows were not persisting. After several consultations, it was determined that the problem was not due to changes in management and that the ration was properly balanced according to traditional chemical measures. In situ analysis showed the rate of corn silage NDF digestion to be .62% h-1, rather than the 4.0% h-1 value found in the CNCPS. The corn silage in the ration contained 35.7% NDF but only 17% of the NDF was digestible at a 3% rate of passage. As a result, a bunk mix designed to support 75 lbs. of milk was predicted to supply only enough metabolizable energy to support 60 lbs. of milk without substantial weight loss. Both the initial and modified rations are presented in Table 4.
To correct the observed ration problems, the diet was rebalanced with the CNCPS. Ration forage was reduced from 63 to 49% of the ration dry matter. Ration starch and fermentable starch were reduced slightly to offset loss of forage dry matter. Fermentable NDF content of the diet increased from 2.6 to 4 lbs. Dietary fermentable carbohydrate increased by 1.7 lbs. Proportion of fermentable carbohydrate provided by fiber increased from 14.9 to 20.7%. As a result of these changes, metabolizable energy available for milk production (ME allowable milk) increased to 69 lbs. per cow per day. These changes allowed cows to increase milk production by 8 lbs. per head per day in less than a week.
Both authors have had similar results in other herds, although circumstances varied. We will summarize our experiences in the following paragraph. Generally, correcting problems with low digestibility forages will increase milk production; the amount of time required for the response depends on the days in milk of the herd. Even in stale herds, it is important to correct poor forage digestibility problems to ensure adequate weight gain for the next lactation. This is especially true in herds using BST. Fermentable fiber sources used successfully have included grass forages, soyhulls, corn gluten feed, brewers grains and beet pulp. Remember to drop ration starch or NFC levels and ensure adequate effective fiber when fermentable byproduct fiber sources replace forages. In some cases, it is not possible to completely offset problems in NDF digestibility because of effective fiber concerns. Corn silage with poor starch (NFC) availability can be corrected by simply increasing corn grain levels. In forages which have poor digestibility of both NDF and NFC (often harvested late), beet pulp and a combination of molasses products and soyhulls have been used to correct the problem successfully. Forages which have poor NDF digestibility due to a lag in NDF digestion (no NDF digestion occurs until several hours after eating), often cause a dual problem by reducing intake levels and providing inadequate amounts of digestible nutrients. Finally, and most importantly, the source of the digestibility problems (harvest management, environment, hybrid selection, etc.) should be identified if possible to avoid or reduce problems in the future.
Conclusions
Digestibility of feedstuffs is a major determinant of their energy content. By measuring feedstuff digestibility and incorporating this information in an appropriate ration balancing package, it is possible to better describe the energy (in)sufficiency of rations.
A number of methods are available for measuring digestibility. In situ digestibility measures provide digestibility values generated under conditions which most closely mimic those a ration component will be exposed to. Both precision and accuracy are concerns with all method used to measure feedstuff digestibility. Measurement of the rate of digestion is not possible with all methods, but appears to be quite important in determining the energy available to the animal.
In situ digestibility data has been used successfully to balance rations over the past two years. This data is used, in concept, much like other forage analysis results. Deficiencies in digestible nutrient content of a forage must be offset by increasing the levels of digestible nutrients provided by other ration components. This can be another forage or a byproduct fiber source in the case of low digestible NDF content or a combination of fermentable starch and fiber sources when both digestible NDF and NFC contents are low. Production responses are generally rapid, unless the herd is quite stale, and have ranged from 3 to 10 lbs. of milk per cow on a daily basis.
References
Ellis, W. C., J. H. Matis, T. M. Hill, and M. R. Murphy. 1994. Methodology for estimating digestion and passage kinetics of forages. p. 682-756. In G. C. Fahey, Jr., M. Collins, D. R. Mertens, and L. E. Moser (ed.) Forage Quality, Evaluation, and Utilization, Madison, WI, USA.
Moore, J. A., M. H. Poore, and R. S. Swingle. 1989. Calculated ruminal extent of digestion as influenced by in situ particle size, forage dosing time, and sampling site in Holstein cows. J. Anim. Sci. 67(Suppl. 1):554(Abstr.).
Siciliano-Jones, J. and N. St. Pierre. 1997. Using in situ data in ration formulation. Proceedings of the TriState Nutrition Conference.
Smith, N. E., M. D. Hanigan, and D. C. Weakley. 1996. Dairy energy systems: Goals, present status, future directions. J. Dairy Sci. 79(Suppl.1):196(abstr.).
Van Soest, P. J. 1996. Environment and forage quality. In Proceedings of the Seeds of Animal Nutrition Symposium, Rochester, NY, USA.
Weiss, W. P. 1994. Estimation of digestibility of forages by laboratory methods. p. 644-681. In G. C. Fahey, Jr., M. Collins, D. R. Mertens, and L. E. Moser (ed.) Forage Quality, Evaluation, and Utilization, Madison, WI, USA.
Weiss, W. P. 1996. Estimating net energy of feeds. J. Dairy Sci. 79(Suppl.1):196(abstr.).
Table 1. Corn silage digestibility parameters, farm samples of 1995 harvest.

1Digestibility of nutrients was determined using the equation: Digestible nutrient = kd/(kd+kp), where is kd rate of digestion of the nutrient and kp is rate of passage out of the rumen.
2Digestible nutrient (discounted for rate of passage) expressed as a percent of the dry matter.
3ND: pool was not detected.
Table 2. Digestibility of corn silage samples from three Eastern New York farms which vary in soil drainage.

1Digestibility of nutrients was determined using the equation: Digestible nutrient = kd/(kd+kp), where is kd rate of digestion of the nutrient and kp is rate of passage out of the rumen.
Table 3. Change in calculated metabolizable energy, Mcal/lb DM, with either a 20% increase or a 20% decrease in size of the indigestible neutral detergent fiber pool or rate of digestion for the digestible neutral detergent fiber pool.
Change in value |
|||
| Forage | Parameter |
20% increase |
20% decrease |
| Corn silage, 50% grain | Indigestible NDF | -.01 |
+.01 |
| Corn silage, 50% grain | Rate of NDF digestion | +.02 |
-.02 |
| Mixed alfalfa/grass silage | Indigestible NDF | -.03 |
+.03 |
| Mixed alfalfa/grass silage | Rate of NDF digestion | +.01 |
-.03 |
| Grass silage | Indigestible NDF | -.01 |
+.01 |
| Grass silage | Rate of NDF digestion | +.02 |
-.02 |
Table 4. Ingredient composition, %DM, of initial and modified rations for example herd.
| Ingredient | Initial Ration |
Modified Ration |
| Soybean meal, 48% | 4.6 |
7.65 |
| Soy Hulls | 0 |
8.7 |
| Prime 32 | 3.04 |
1.45 |
| Mineral pack | 2.26 |
2.16 |
| Whole cottonseed | 4.96 |
2.09 |
| Protein mix | 1.24 |
.86 |
| Corn meal | 0 |
6.54 |
| Alfalfa silage, 17% | 21.37 |
17.05 |
| High moisture corn | 11.88 |
11.46 |
| Limestone | .47 |
0 |
| Distillers grains, dried | 7.37 |
10.34 |
| SoyPlus | 1.24 |
0 |
| Megalac | 0 |
0 |
| Corn silage | 41.58 |
27.64 |
| Grass hay, 11% CP | 0 |
4.05 |